Mistletoe Clinical Trial
Phase I Complete. Phase II Underway This Year.
Believe Big is honored to be on the front lines of groundbreaking cancer research. In partnership with Johns Hopkins University School of Medicine, we are helping lead the first U.S. clinical trial using intravenous mistletoe, bridging the gap between conventional and complementary medicine in the fight against cancer.
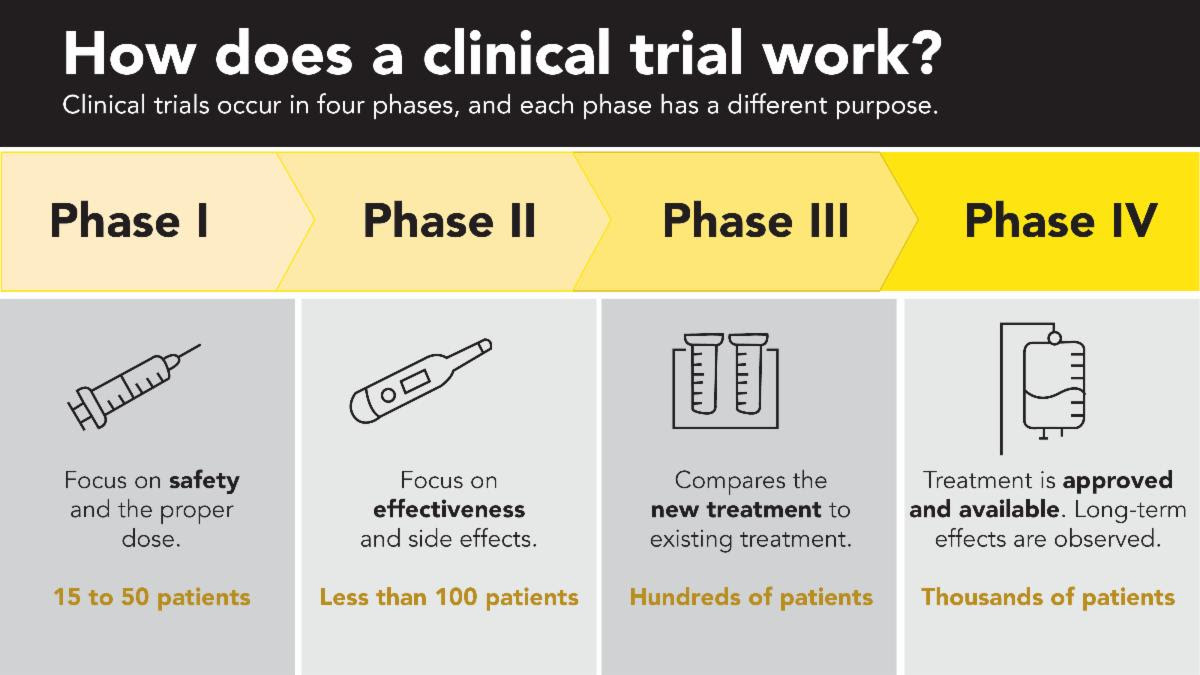
While mistletoe therapy has been widely used in countries like Germany and Switzerland for over 100 years, bringing it into standard oncology care in the United States requires rigorous clinical trials. Each phase builds on the last, answering critical questions about safety, effectiveness, and real-world impact.
We are thrilled to share that Phase I is complete, demonstrating that mistletoe is safe and showing promising potential to improve patients’ quality of life. Phase II is set to begin in the coming months, bringing us one step closer to the answers that could change the standard of care for cancer patients across the country.
But the journey does not end there.Phase III will be the pivotal next step that can open the door to widespread adoption and insurance coverage, making this therapy accessible to patients everywhere. And unlike conventional treatments backed by major pharmaceutical funding, this research depends on a community of passionate supporters.
That’s where you come in.Your support helps move this research forward, bringing hope, expanding treatment options, and changing lives.
Download and read the published study here.
To find out more about Mistletoe in science and medicine click here.
Help Bring Phase II to Life!
As Dr. Diaz shared, Phase I was like scoring a touchdown in the Super Bowl—an extraordinary win. Now, with Phase II underway, we’re driving down the field with purpose and momentum. But we’re not done yet.
We need to finish strong.
Your support today helps carry this research across the finish line.
Together, we can make Phase II a reality.
Let’s Get Phase II Funded
Early results are promising! Phase I showed that mistletoe is safe and may significantly improve patients’ quality of life. Together, we can take the next bold step.
Your support can help bring Phase II to life! Advancing this research, expanding hope, and impacting countless lives.
Believe Big only allows 5 percent on overhead of institutional funding.
LEGAL DISCLAIMER
FDA Required Disclaimer For Sites That Do Not Endorse Chemotherapy:
This web site is for educational purposes only. It is not intended as a substitute for the diagnosis, treatment and advice of a qualified licensed professional. This site offers people medical information beyond conventional medical options, but in no way should anyone consider that this site represents the “practice of medicine.” This site assumes no responsibility for how this material is used. Also note that this website frequently updates its contents, due to a variety of reasons, therefore, some information may be out of date. The statements regarding medical information beyond conventional treatments for cancer have not been evaluated by the FDA